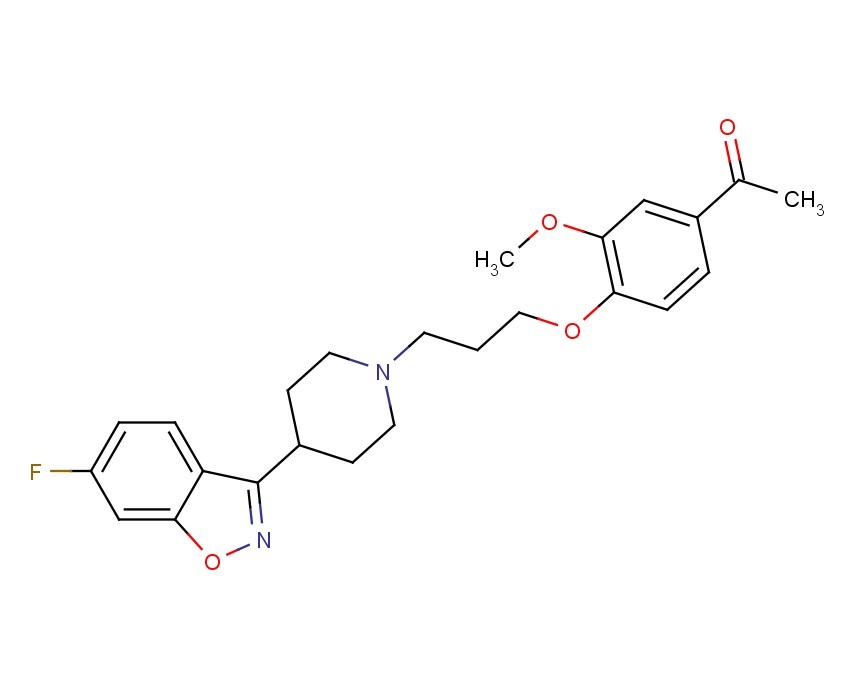
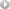BMS-790052 is a highly selective inhibitor of HCV NS5A with EC50 of 9-50 pM, for a broad range of HCV replicon genotypes and the JFH-1 genotype 2a infectious virus in cell culture.
IC50 Value: 9-50 pM
Target: HCV NS5A
BMS-790052 has broad genotype coverage and exhibits picomolar in vitro potency against genotypes 1a (EC50 50pm) and 1b (EC50 9pm). BMS-790052 produces a robust decline in HCV RNA (-3.6 logs after 48 hours from a single 100 mg) dosefollowing a single dose in patients chronically infected with HCV genotype 1.
[1]. Fridell RA et al. Resistance analysis of the hepatitis C virus NS5A inhibitor BMS-790052 in an in vitro replicon system. Antimicrob Agents Chemother. 2010 Sep;54(9):3641-50.
Abstract
BMS-790052 is the most potent hepatitis C virus (HCV) inhibitor reported to date, with 50% effective concentrations (EC(50)s) of < or = 50 pM against genotype 1 replicons. This exceptional potency translated to rapid viral load declines in a phase I clinical study. By targeting NS5A, BMS-790052 is distinct from most HCV inhibitors in clinical evaluation. As an initial step toward correlating in vitro and in vivo resistances, multiple cell lines and selective pressures were used to identify BMS-790052-resistant variants in genotype 1 replicons. Similarities and differences were observed between genotypes 1a and 1b. For genotype 1b, L31F/V, P32L, and Y93H/N were identified as primary resistance mutations. L23F, R30Q, and P58S acted as secondary resistance substitutions, enhancing the resistance of primary mutations but themselves not conferring resistance....
[2]. Suzuki F, Sezaki H, Akuta N, Suzuki Y, Seko Y, Kawamura Y, Hosaka T, Kobayashi M, Saito S, Arase Y, Ikeda K, Kobayashi M, Mineta R, Watahiki S, Miyakawa Y, Kumada H.Prevalence of hepatitis C virus variants resistant to NS3 protease inhibitors or the NS5A inhibitor (BMS-790052) in hepatitis patients with genotype 1b.J Clin Virol. 2012 Aug;54(4):352-4. Epub 2012 Jun 1.
Abstract
BACKGROUND: Hepatitis C virus (HCV) of genotype 1b is the most prevalent worldwide, and the least responsive to interferon-based treatments. A combination therapy with two direct-acting antivirals has shown promising results in patients with HCV-1b, but the prevalence of drug-resistant variants before treatment is not known in the Japanese population. OBJECTIVES: To detect HCV variants resistant to NS3 protease inhibitors or the NS5A inhibitor (BMS-790052) in hepatitis patients infected with HCV-1b. STUDY DESIGN: Drug-resistant mutations were determined in the 362 hepatitis patients infected with HCV-1b who had not received direct-acting antivirals before. RESULTS: Amino-acid substitutions resistant to NS3 inhibitors (V36A, T54S, Q80H and D168E) were detected in 15 of the 307 (4.9%) patients, who had been examined, and T54S (3.3%) predominated over V36A (0.3%), Q80R (0.7%) and D168E (0.7%) in them. Amino-acid substitutions resistant to BMS-790052 (L31M and/or Y93H) were detected in 33 of the 294 (11.2%) patients, and Y93H (8.2%) predominated over L31M (2.7%). One of the 239 (0.4%) patients, who had been examined for amino-acid substitutions in both NS3 and NS5A regions, possessed HCV-1b variants resistant to NS3 inhibitors (T54S) and BMS-790052 (L31M)...
[3]. Jiang H, Zeng J, Kandoussi H, Liu Y, Wang X, Bifano M, Cojocaru L, Ryan J, Arnold ME.A sensitive and accurate liquid chromatography-tandem mass spectrometry method for quantitative determination of the novel hepatitis C NS5A inhibitor BMS-790052 (daclastasvir) in human plasma and urine.J Chromatogr A. 2012 Jul 6;1245:117-21. Epub 2012 May 14.
Abstract
A liquid-liquid extraction (LLE) and liquid chromatography-tandem mass spectrometry (LC-MS/MS) methods have been developed and validated for the quantification of BMS-790052 (daclastasvir) in human plasma and urine. The samples were extracted with methyl-t-butyl ether (MTBE) before analyzing by an API 4000 mass spectrometer which was operated in a multiple reaction monitoring (MRM) mode for detection of positively charged ions of BMS-790052 and its internal standard, 13C??-BMS-790052. The standard curves ranged from 0.050 to 50.0 ng/mL for BMS-790052 in human plasma, and 1.00-1000 ng/mL in human urine. The intra-assay precision (%CV), based on four levels of analytical QCs (low, geometric mean, mid and high), was within 8.6%; inter-assay precision (%CV) was within 6.7% for both plasma and urine methods, and the mean assay accuracy (%Dev) was within ±3.0% for both plasma and urine methods. The ruggedness of this accurate, precise, and selective LLE-LC-MS/MS method has been demonstrated in the successful analysis of several thousand clinical study samples.
[4]. Sun JH, O'Boyle Ii DR, Zhang Y, Wang C, Nower P, Valera L, Roberts S, Nettles RE, Fridell RA, Gao M.Impact of a baseline polymorphism on the emergence of resistance to the hepatitis C virus nonstructural protein 5A replication complex inhibitor, BMS-790052.Hepatology. 2012 Jun;55(6):1692-9.
Abstract
The influence of naturally occurring polymorphisms on the potency of the HCV nonstructural protein 5A (NS5A) replication complex inhibitor, BMS-790052, was investigated by evaluating hybrid replicons in which the entire NS5A coding region of genotype (GT) la and 1b laboratory (lab) strains (H77c and Con1) were replaced with the corresponding regions of specimens collected from 10 GT-1a- and 6 GT-1b-infected subjects. For baseline (BL) specimens, with no previously observed resistance variants identified by population sequencing, the median 50% effective concentration (EC(50) ) values for BMS-790052 were similar for the clinically derived and lab strains. A Q30R variant was observed at viral breakthrough (VBT) in one of the GT-1a-infected subjects. Because the lowest plasma exposure of BMS-790052 observed in this subject was 117 nM and the median 50% effective concentration value for a GT-1a H77c replicon containing a Q30R substitution is ~7 nM, a rigorous investigation was initiated to determine the basis for resistance. Three approaches were used: (1) replacement of the entire H77c NS5A or (2) replacement of the N-terminal region of NS5A, with sequence from BL and day 14, and (3) substitution of specific amino acids. A BL polymorphism (E62D) did not contribute resistance to BMS-790052; however, the linked variant, Q30R-E62D, conferred high-level resistance in vitro and is likely responsible for VBT in vivo. CONCLUSION: Our data show that a BL polymorphism with minimal effect on the anti-HCV effect of BMS-790052 can affect the emergence of resistance and significantly affect clinical outcome. This work establishes a clear, systematic approach to monitor resistance to NS5A inhibitors in the clinic.
[5]. Wang C, Jia L, Huang H, Qiu D, Valera L, Huang X, Sun JH, Nower PT, O'Boyle DR 2nd, Gao M, Fridell RA.In vitro activity of BMS-790052 on hepatitis C virus genotype 4 NS5A.Antimicrob Agents Chemother. 2012 Mar;56(3):1588-90. Epub 2011 Dec 27.
Abstract
The antiviral profile of BMS-790052, a potent hepatitis C virus (HCV) replication complex inhibitor targeting nonstructural protein NS5A, is well characterized for HCV genotype-1. Here, we report that BMS-790052 inhibits hybrid replicons containing HCV genotype-4 NS5A genes with 50% effective concentrations (EC(50)s) ranging from 7 to 13 pM. NS5A residue 30 was an important site for BMS-790052-selected resistance in the hybrid replicons. Our results support the potential of BMS-790052 as a valuable component of combination therapy for HCV genotype-4 chronic infection.







 Antibody-drug Conjugate/ADC Related
Antibody-drug Conjugate/ADC Related